Congratulations to this year’s winners and shortlisted entries for the HSDTC Public Engagement Competitions!
Doctoral researchers in the Health Sciences submitted entries for the HSDTC Science Communication Prize and the HSDTC Science Image Prize for a chance to win £300 and be featured on our website and blog. Keep reading to learn more about the shortlisted entries for 2022.
HSDTC Science Image Prize 2022
Doctoral researchers had to submit a visual entry that either conveyed the entirety of their research or an aspect of it.
We were looking for images which might explore the social impact that their research has on a global, population, or individual scale. They may be a true representation of the subject, or manipulated post-production, such as with false colouring; or they could show their research in a more abstract way.
Images could have shown, for example, the subject matter of their research, such as an image of cells or the brain, or research in action, for example the methods, equipment or facilities used in their research.
The judges were looking for images which are informative, captivating, and which clearly communicate the research being done. Entries could utilise many techniques, such as: photography; microscopy; medical imaging; new and emerging imaging techniques; data visualisation; artistic media.
The impactful shortlisted submissions are available below, including our winner Antzela Tsavou, and runner-up, Natalie Sanford:
Antzela Tsavou (winner)
Host-Microbiome Interactions, Faculty of Dentistry, Oral & Craniofacial Sciences
Yin and Yang

Yin and Yang by Antzela Tsavou
Life is full of opposites. Completely contrary forces that we think do not interrelate at all. Yet, instead of being all black or all white, it’s a little of both, just like Yin and Yang. The fungus that I study, Candida albicans, is a pathobiont which means that it exists as a helpful member of the human microbiota until the opportunity is found to be harmful in immunocompromised people. This fungus can switch reversibly between “the good” or yeast form (on the left) and “the bad” or hyphal form (on the right) to take control of the human host and manipulate it just like a master of survival.
Two worlds that give rise to each other. Just like in life and in nature, maintaining balance or the so-called homeostasis is key. Evolution has allowed this microorganism to control the human host and transition between these two very different worlds with ease.
In my PhD, I study the control of this morphological switch. Future work towards identifying additional regulators of this change could open up new avenues towards treatment developments.
These images were taken using the EVOS M5000 microscope and contrast was manipulated using Photoshop.
Natalie Sanford (runner-up),
Methodologies Division, Florence Nightingale Faculty of Nursing, Midwifery & Palliative Care
Pressure

A water colour painting depicting hands lifting or supporting an exhausted healthcare provider, who is overwhelmed with a variety of competing priorities. Photo of painting enhanced to suit digital format.
Stijn Aaron den Daas (shortlisted)
School of Immunology & Microbial Sciences, Faculty of Life Sciences & Medicine
Heart Organoids
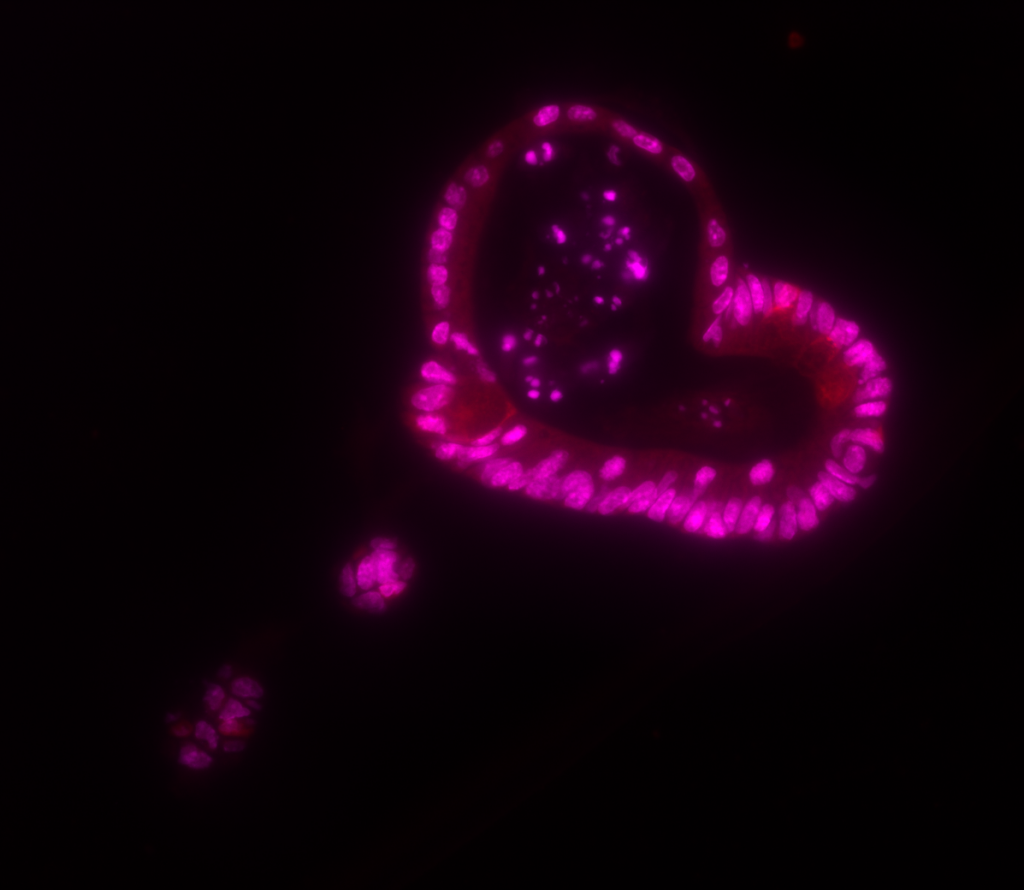
A heart shaped intestinal organoid stained for Lysozyme (Red) and nuclear counterstain (Magenta), imaged with a fluorescent microscope, and coloured with ImageJ. Intestinal organoids are grown from isolated intestinal crypt cells and grown in specialised media to allow the crypt stem cell to proliferate, while maintaining their stem-ness. Cells proliferated from stem cells differentiate into the various cell types present in the intestinal epithelium, i.e. Enterocytes, Paneth cells, Goblet cells, Tuft cells, and Enteroendocrine cells. These cells form an intact monolayer with the apical side inward and basolateral side outward, creating the lumen inside the intestinal organoid. While growing the organoids create crypt and villi –like structures that resemble the intestine, which result in a 3D budding spherical shape.
We use intestinal organoids as a model for the intestine to translate disease aetiology of liver cirrhosis, with a focus on the gut-liver axis in disease progression. Intestinal organoids can be used to examine the effect of toxins and therapies on the intestinal epithelium in vitro.
Zsofia Szlamka (shortlisted)
Psychology, Institute of Psychiatry, Psychology & Neuroscience
Autism-friendly

The subject of this image is a world that is entirely autism-friendly and inclusive. While the blue context calls for autism, the nature represents the entirety, the Globe itself. The picture was taken with a smart phone and no post-production manipulation took place.
Anna Elisabeth Fürtjes (shortlisted)
Social, Genetic & Developmental Psychiatry Centre, Institute of Psychiatry, Psychology & Neuroscience
Pursuing a PhD

The image I am submitting to the HSDTC Science Image Competition depicts 3-dimensional coordinates of a standard human brain with random colours spread across the cortex. I have produced the brain images while figuring out how to use an algorithm to subdivide the brain into contiguous areas. My first attempts resulted in images that looked like the brain on the bottom left. Through trial and error, I started to understand the inner workings of the algorithm, and I managed to manipulate it in a way that it allowed me to produce the brain on the right depicting contiguous areas, as I needed them for my study. This process was spread across a few weeks as it was time-consuming and computationally demanding.
The image represents the process of Pursuing a PhD as I have experienced it throughout the past 2 ½ years. I learned that conducting research is never straightforward, and using trial and error helped me get better at coding to manipulate datasets allowing me to answer niche biological questions about the brain.
The brain images were generated with the programming language R, and I used specialised packages within R (Rvcg & rgl) to render the images from 3-dimensional coordinates. The 3D coordinates are commonly used by neuroscientists as a reference, when pre-processing their brain images (fsaverage). I assembled the brain renders created with R in Adobe Photoshop. The R code depicted in the background of the final image is taken from the R scripts I wrote to generate the brain renders.
HSDTC Science Communication Prize 2022
PhD students submitted short ‘newspaper style’ articles about research projects they were currently involved in (or an aspect of it). The articles are aimed at lay audiences and have to be interesting and understandable for an interested member of the public.
The judges were looking for articles which:
- are compelling to read and easily understandable
- clearly explain the research being done
- answer the question “why does this research matter?”
- are worthy of publication in a national newspaper.
Five engaging submissions were shortlisted, including the winning submission by Lea Lortal, and the runner-up, Oscar James MacCormac:
Lea Lortal (winner)
Centre for Host-Microbiome Interactions, Faculty of Dentistry, Oral & Craniofacial Sciences
When your own fungus betrays you
The fungi kingdom is very broad: it ranges from very big mushrooms to microscopic yeasts. What’s peculiar is that some of these fungi are friendly as they are used for food and medicine, but others are hostile and cause disease in humans.
Fungal infections are incredibly common: they affect 1 billion people around the world. That’s 1 in 8 people.
Some of these infections are superficial (e.g. nail infections) but others are life-threatening (e.g. blood infections) and kill 1.5 million people every year.
Despite these high numbers, this mysterious kingdom has been neglected, and remains understudied and underfunded.
One of the most common pathogenic fungi is Candida albicans. This yeast is naturally present in our body and is usually harmless. But in specific conditions, it can overgrow and shift to a harmful version of itself, causing infections such as thrush. Talk about a betrayal.
In 2016, the Naglik lab at King’s College London discovered that when Candida albicans transitions from friendly to harmful, it produces a toxin. They named it ‘candidalysin’.
Today, the Naglik & Richardson labs are leading the effort to understand how candidalysin physically and chemically damages human cells. “By investigating how candidalysin works, our goal is to enhance our understanding of Candida albicans’ ability to cause disease and contribute to the identification of new therapeutic targets.” says PhD candidate Antzela Tsavou.
The discovery of candidalysin shows that the fungal kingdom is still full of mysteries. In the last two years, we have witnessed the emergence of Covid-19-associated fungal infections, with alarmingly high mortality rates. Fungal infections are on the rise, and their studying is a moral imperative for this next decade.
Oscar James MacCormac (runner-up)
Biomedical Engineering and Imaging Sciences, Faculty of Life Sciences & Medicine
Hyperspectral, Hypersafe!
A research group at King’s College Hospital are currently developing a novel imaging system for use during surgery in order to improve the efficiency and safety of brain tumour surgery. The theory behind this is that the human eye, along with our best smartphone cameras, is only capable of seeing colours within the red, green and blue light ranges; hyperspectral cameras allow us to detect far greater detail than these colours alone. To explain this further, Anisha Bahl; one of the PhD students working with the project says ‘imagine seeing a selection of apples in the supermarket. All of them look broadly the same, however when you take a bite the apple may be bruised inside. The hyperspectral camera allows us to see the bruised apples before we leave the supermarket.’
The clinical use of this technology is becoming increasingly clear and exciting; applying the above analogy, this will allow neurosurgeons to see, live, clearly what is tumour tissue and what is not meaning more precise, safer surgery for patients. On top of this, no radiation is involved – the system relies solely on simple white light, meaning no additional risk for any patients undergoing surgery with this revolutionary new technology.
Patients who have been through brain tumour surgery are also enthusiastic about this prospect; patient representatives from patient and public involvement (PPI) group for the project have said ‘any opportunity to make brain surgery safer for patients is an exciting prospect’ and ‘our first PPI group session provided a great insight in to the views of others and provoked some fascinating discussion regarding the role of artificial intelligence in surgery – actually very well received as an aid to guiding the surgeon.’
The National Institute for Health and Care Research (NIHR) agrees with the above, awarding the group a coveted i4i research grant in order to run the first clinical trial of the system at King’s College Hospital due to commence in May 2022; we look forward to keeping you updated with its progress!
Natalie Sanford (shortlisted)
Methodologies Division, Florence Nightingale Faculty of Nursing, Midwifery & Palliative Care
Creating the Perfect Harmony: Interprofessional Teamwork in Healthcare
Quality patient care depends on healthcare workers from many disciplines working together seamlessly –teamwork which requires cross-disciplinary understanding, coordination, and communication – yet there is currently no international standard for teaching this critical skillset. Researchers at King’s College London are working to change that, starting by evaluating the current state of teamwork in hospitals.
A healthcare team is not unlike an orchestra made up of many different, complementary instruments. While each section rehearses separately, the full ensemble must practice together before opening night. Yet, in healthcare, where the stakes are quite literally life and death, the full team does not always rehearse together before being expected to perform. Becoming a healthcare provider in any discipline requires substantial training, yet there are currently no international requirements for interprofessional education. When the curtain goes up, it’s assumed that everyone will play their parts.
A team of King’s College London researchers are analysing teamwork in hospitals in England to better understand how different professions are already working together. The hope is that evaluating how teamworking takes place organically might offer insight into how education can be improved and what support workers need to be more effective in their roles. The challenges and pressures healthcare workers face are extreme and have been exacerbated by the COVID-19 pandemic through decreased staffing, sicker patients, and fewer resources. The research team is examining these challenges and pressures, observing how workers flexibly adapt to meet demands despite reduced capacity, and whether this differs based on the features of the team. The setting, structure, membership, and function of the team are all factors that might influence how teams work together to coordinate their actions, researchers have found. Understanding what constitutes successful teamwork can enable researchers to devise ways of training and supporting teams to increase patient safety, reduce burnout, improve job satisfaction, and retain the existing healthcare workforce. Whether seasoned performers or newcomers to the hospital stage, the show must go on – and our healthcare workforce deserves a dress rehearsal in addition to broken legs!
Samia Elkommos (shortlisted)
Basic and Clinical Neuroscience, Institute of Psychiatry, Psychology & Neuroscience
Solving the Seizure Puzzle – Is Wearable Technology the Missing Piece?
Seizures happen for many reasons – but trying to find out the type of seizure, as well as the trigger, can be a puzzle and take some detective work. Often, doctors use lots of pieces of information to work this out. They’ll assess the person’s account of what happened, any witnesses who saw the seizure, a video if available, and carry out medical tests. None of these are 100% reliable though, and even after extensive research it can be difficult to know what type of seizure someone has had.
A common type of seizure is an epileptic seizure which occurs due to abnormal electrical activity in the brain. At the opposite end of the spectrum, functional (non-epileptic) seizures are thought to occur due to dissociation – where people experience a disconnect between their thoughts, surroundings, and identity.
It is important to differentiate between the two because their treatments are completely different – epilepsy is treated with medication, whereas functional seizures are treated with talking therapies. People can get hurt if they are diagnosed incorrectly and receive the wrong treatment. To the naked eye both seizures look similar – so it can be tricky.
Wearable technology is being used in epilepsy research, and shows that changes in the body which occur around the time of seizures can be measured with wearable sensors worn on the wrist or arm. By measuring changes in heart rate, arm movements, breathing and skin sweating we can detect when someone has had an epileptic seizure.
We theorise there will be different patterns in heart rate, arm movements and breathing between those with epileptic seizures and those with functional seizures. As such, wearable technology could be the missing piece of the puzzle, as it gives real-time information about what is happening to the body around the time of seizures.
If we can use this to identify different patterns of body changes in epileptic seizures versus functional seizures we can diagnose patients more quickly, more accurately and start them on the correct treatment from day one.
Lucy Menage (shortlisted)
Basic and Clinical Neuroscience, Institute of Psychiatry, Psychology & Neuroscience
Taming the beast: taking the bite out of rabies virus
Walking down a dark and desolate street, the eerie still of the night is broken by the soft patter of footsteps, but they are not yours. You look around you in a panic trying to locate the source. Suddenly, a dog reveals itself from the shadows, lips ferociously snarled around its teeth and producing a low, bloodcurdling growl from deep within its throat. You gulp as you notice long strands of saliva dripping from the beast’s mouth. The dog braces itself, ready to pounce.
A scene such as this would have terrified someone in the 19th century as it signaled a dog carrying canine madness or what we know today as the rabies virus. Rabies is a horrifying virus able to induce peculiar symptoms such as extreme aggression, insomnia, and aversion to light, leading some to suggest rabies as the origin of the vampire myth.
The success of rabies comes from its ability to pass from the initial wound of entry all the way to the brain in a backwards, or retrograde, manner. This amazing ability has since been harnessed for neuroscience. Brain cells form connections with each other to allow communication between different regions. For example, the ‘I’m hungry’ region needs to speak to the ‘go to the fridge’ region. By meticulously altering the rabies virus, scientists have transformed it into a powerful tool for mapping these connections. Modified rabies, which now emits a colourful green light, is injected into a brain cell. The infective nature of rabies means it spreads backwards to an input cell, still emitting that green light. However, the tweaked virus can move no further and remains in the first connection. Why is this important? Well, the first connection indicates a strong cellular conversation and seeing how neurons communicate with each other is crucial to understanding their function within the brain. Once we understand the function, we’re one step closer to understanding the dysfunction in brain diseases. This is just one example of why science is so exhilarating, being able to convert a devastating disease into a tool for improving our future health.

















